Is your organization ready
for the new 2016/2017 CMS Quality Reporting programs?
Increase Quality Assurance for your organization's submissions
to the new CMS QRDA compliance programs today!
Our clients have leveraged our
QRDA support to expedite their 2016/2017 reporting processes
to Federal MIPS/APS/CPC+, State Medicaid, payer and internal QA initiatives
|
|
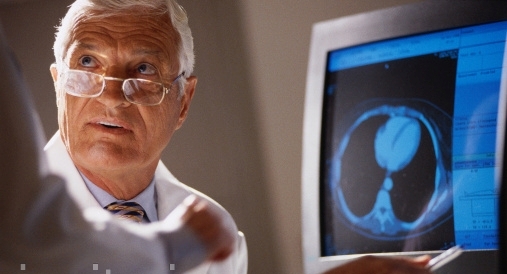 |
Minimize CMS penalties and
maximize incentives
for your
provider organization
with our CMS QRDA submission review tools
Optimize your CMS incentives for
your providers by using our proprietary QRDA submission review tools,
and minimize the penalties and workflow interruptions to your providers.
View MIPS / APS / CPC+ tools here.
|
|
QRDA-HISP supports
your organization |
Expedite your
CMS report process
with our QRDA modules
QRDA Solutions provides
modular QRDA
support through our secure cloud to supply the QA report tools
your organization needs.
When you simply your process by
using our bolt-on QRDA toolkit, your
organization can create submissions for CMS EH and EP organizations and State QA
initiatives by exporting patient data from
your EHR.
|
|
QRDA services
Improve submissions
Lower costs
CMS & ONC compliance
|
We lower your CMS reporting costs
Registries and EHR vendors
charge a premium for QRDA report services; many report processes can
be performed by your IT staff.
Does your organization need to
improve the QRDA submission while containing QRDA reporting costs?
Our
QRDA toolkit eliminates the
CMS and QRDA related charges for EHR modules or retain expensive registry
consultants, who are simply translating your exported patient data.
|
|
|
 |
Our process leverages your current
technology.
Problem: Your current EHR
version does not fulfill CMS requirements, while your EHR
vendor requires costly upgrades that may break or change other EHR
functions.
Solution: Our procedures
leverage your current technology to optimize CMS measure selection
for all applicable CMS reporting channels achieve 2016 & 2017 CMS
submission compliance.
Benefit: IT requirements are minimized
for 2016 & 2017 CMS requirements.
|
|
|

|
We leverage your current IT resources
Solution: Your IT staff
knows how to extract reports from your EHR.
Why pay an expensive registry to duplicate these efforts?
Benefit: Staff resources are used instead of hiring an
expensive CMS Registry consultant.
|
|
|
 |
Our processes fulfill CMS QRDA report requirements
and minimize workflow impact
Problem: New CMS 2015 mandates
triple the payload required of CMS submissions.
Solution: Our procedures
streamline CMS report logistics to optimize
all stages of QCDR submission preparation.
Benefit: Your practice's CMS
payment adjustments are optimized with minimum workflow
interruptions.
|
|
|

|
Scheduled automated validation
of your CMS submissions
Solution: Our Submission
Auditor uses proven data validation and scrubbing processes
where errors are identified at all stages of the CMS / QRDA
submission
generation process. Our CMS
Measure Map Compiler extracts strategic data from your EHR, PM,
and CMS
billing records to optimize CMS measure selection and evaluating
CMS quality report channels.
Benefit: Validated CMS reports save time and resources for your staff.
|
|
|
CMS QCDR
Compliance
Audit |
|
 Passed
Passed |
|
Our Dashboard manages the QCDR/QRDA submission process
Solution: Our dashboard
manages the generation, review, and transmittal of complex QRDA
documents sourced by multiple EP electronic record systems by
leveraging proven financial audit trail technologies.
Benefit: Our dashboard enables efficient management
of the CMS submission process.
|
|
QRDA
Quality
Reporting
Document
Architecture
CMS
2015 QRDA specs |
cc: To all Healthcare Providers and HIT Vendors:
2016 and 2017 CMS rules change your submissions...
CMS mandates QRDA compliance
Is your organization
ready for QRDA?
CMS 2016 rules mandate Quality
Report Document Architecture (QRDA) compliance for quality
submissions from CMS healthcare delivery systems.
When is your first QRDA
submission due?
We expedite your capacity to
generate, validate and review QRDA submissions by
having tested solutions that extends life cycle of your existing
systems.
Read more...
|
|
|
 |
Is your organization installing redundant Quality
Assurance (QA)
systems?
After installing multiple
QA report subsystems that are probably redundant, many Healthcare
organizations are suffering these symptoms:
- Exploding CMS quality reporting costs - MIPS/APS/CPC+/QRDA/VBM...
- Escalating maintenance upgrades
- Lack of CMS compliance - Missed submission
dates
Multiple QA installations cause
these problems when vendors can not meet inevitable Federal and
State regulatory timetables for the innumerable changes in
interrelated submission requirements.
Our QRDA toolkit minimize the
resources, complexity and time required by centralizing Federal
(CMS/CDC) and State QA functions Read
more . . .
|
|
 |
QCDR has already used our QRDA report subsystems
CMS designated the Qualified
Clinical Data Registry as the CMS report channel to test QRDA
report processes.
Healthcare organizations and
QCDR's have discovered that the increased CMS submission payload
increases the development and support costs for QA report subsystems
can
dramatically escalate if universal QRDA subsystems modules are not
implemented.
We have spearheaded the
outsourcing of QRDA compilers and quality report applications.
Our system components encapsulate regulatory updates; This enhances
the value of QRDA to your organization.
Our concise integration of QRDA modules significantly reduces the
time and costs of development, maintenance and future support.
Read more . . .
|
|
|
|
|
|
QRDA
review
Checklist
Eligible measure list
Data integrity
Vetting
|
Our QRDA review tools minimize regulatory exposure
We provide the
tools necessary to review QRDA reports before they are transmitted
to regulatory agencies.
Our QCDR-HISP
Hybrid Cloud insures that your organization complies with the CMS
mandated CMS record integration processes.
|
|
CMS
MU2
Mandates
Expanded measure list
VBM support
Data integrity
Vetting
|
Our QRDA
modules EHRs comply with MU2
The myriad of MU2 EHR mandates
for CMS Quality Submissions increase development costs and time to
market. 2014 has already initiated another cycle of CMS
CMS rule updates which will propagate into code changes; CMS
functions can be simplified with outsourced functions by using a
systematized approach dedicated to MU2 CMS compliance.
Our QCDR-HISP Hybrid Cloud
insure that your EHR complies with the CMS mandated MU2
reporting requirements.
|
|
QRDA/CMS
Audit support
CMS & HIPAA compliance
ISO-9000 documentation
|
We document and secure your QRDA/CMS audit trails
Documentation is central to
effectively managing IT resources that respond to Federal mandated
report programs; tracking regulatory changes can grow into expensive and time consuming upgrades.
Outsourcing various QRDA & CMS components streamline these
processes.
Our documentation and implementation procedures
insure that you maintain schedule and cost control over the CMS
submission process.
|
|
|
 |
Our QRDA processes plan for MIPS regulatory changes
Your organization can reduce regulatory exposure by embracing modular
solutions using our QCDR-HISP Hybrid Cloud. Following regulatory
trends is embedded in our system design processes.
Documentation is
central to effectively managing IT resources that respond to Federal
mandated report programs; tracking regulatory changes can grow into
expensive and time consuming upgrades. Outsourcing
various QRDA & CMS components streamline these processes.
Our documentation
and implementation procedures insure that you maintain schedule and
cost control over the QCDR submission process.
CMS and many state
health agencies currently require Clinical Quality Reports (CQR) to
be submitted using the Quality Reporting Document Architecture (QRDA).
The CMS audit
process for QCDRs requires the ability to be able to trace
associated CMS and medical records while maintaining HIPAA
compliance. Automating the CMS compliance processes usually
involves the complex integration of CMS-1500/837P claim records.
|
|
We save your organization time, effort and
resources to comply with QRDA standards.
|
|
In order
to fulfill CMS QRDA quality reporting requirements,
CMSGATEWAYS.COM
compiles and validates incoming data from many sources as well as the CMS submissions with our Report Generation Engine
by using our efficient data-mining processes for
provider HIT databases and EHRs.
|
|
|
CMSGATEWAYS.COM
provides a support services
to fully leverage your existing
registry IT to enable your
organization to orchestrate a tiered, efficient QRDA submission process. More... |
|
QRDA - QRDA roundtable at ONC 2014 Meeting Summary report available for
@ 2014 ONC Annual meeting Details
here |
|






